Tartaric Acid BP Ph Eur EP IP JP USP NF Analytical Reagent FCC Food Grade Manufacturers, with SDS MSDS Sheet |
Supplier, Manufacturer, Exporter of Tartaric Acid BP Ph Eur EP JP IP USP NF Analytical Reagent FCC Food Grade, Muby Chemicals of Mubychem Group, established in 1976, is the original manufacturers of Specialty Chemicals, Pharmaceutical Excipient, Fragrance Food & Flavor chemicals, Reagent Grade Chemicals, Shale Gas Fracturing Chemicals in India. Mubychem Group has several manufacturing facilities spread across Western India and world wide contacts and toll manufacturers. We are exporting globally to countries like USA, Canada, Europe, UAE, South Africa, Tanzania, Kenya, Egypt, Nigeria, Cameroon, Uganda, Turkey, Mexico, Brazil, Chile, Argentina, Dubai, Korea, Vietnam, Thailand, Malaysia, Indonesia, Australia, China, Germany, France, Italy, Portugal, Bangladesh, etc. The products are offered as per required specifications and in correct shape and size in mm or meshs or microns as specified by the buyer. The participating units have one or more accreditations like FDA - cGMP and GLP approval, ISO-9001 Certified, "REACH" Registered, ISO-14001, ISO/IEC 17025, ISO-22000, FSSC 22000, ISO 45001, Kosher Certified, Halal Certified, HACCP, FSSAI. We offer Commercial Pure & IP BP EP Ph Eur USP NF JP FCC Food Grade Analytical Reagent Grades of Chemicals |
| Bookmark this Web Site -- or -- Email This Page Info to a Colleague or Yourself |
Search our website here:







Tartaric Acid: CAS Number: 87-69-4, EINECS EC Number: 201-766-0, Molecular Formula: C4H6O6, Molecular Weight: 150.1 HS Code ---**
How big is your requirement or how small
We serve it all.
Specifications, Safety Data Sheet, Manufacturing process details, Wholesale retail buy sell prices, Uses etc available on line in these pages for Tartaric Acid.
For MSDS Sheet Click
MSDS Sheet of Tartaric Acid Manufacturers
Spanish Fabricantes de ácido tartárico
Portuguese Fabricantes de ácido succínico
Tartaric Acid
BP Ph Eur EP JP IP USP FCC Food Grade Suppliers
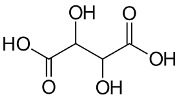
Tartaric Acid BP Ph Eur Grade Specifications
C4H6O6 --- 150.1--- CAS 87-69-4
Ph Eur
DEFINITION
(2R,3R)-2,3-Dihydroxybutanedioic acid.
Content: 99.5 per cent to 101.0 per cent (dried substance).
CHARACTERS
Appearance: White or almost white, crystalline powder or colourless crystals.
Solubility: Very soluble in water, freely soluble in ethanol (96 per cent).
IDENTIFICATION
A. Solution S (see Tests) is strongly acid.
B. It gives the reactions of tartrates.
TESTS
Solution S: Dissolve 5.0 g in distilled water and dilute to 50 ml with the same solvent.
Appearance of solution: Solution S is clear and not more intensely coloured than reference solution.
Specific optical rotation: + 12.0 to + 12.8 (dried substance)
Oxalic acid: Maximum 350 ppm, calculated as anhydrous oxalic acid.
Chlorides: Maximum 100 ppm.
Sulphates: Maximum 150 ppm.
Calcium: Maximum 200 ppm.
Heavy metals: Maximum 10 ppm.
Loss on drying: Maximum 0.2 per cent, determined on 1.000 g by drying in an oven at 105C
Sulphated ash: Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.650 g in 25 ml of water. Titrate with 1 M sodium hydroxide using 0.5 ml of
phenolphthalein solution as indicator, until a pink colour is obtained.
1 ml of 1 M sodium hydroxide is equivalent to 75.05 mg of C4H6O6.
Tartaric Acid USP NF Grade Specifications
C4H6O6 -- 150.09
Butanedioic acid, 2,3-dihydroxy-; Butanedioic acid, 2,3-dihydroxy-, [R-(R*,R*)]-.
Tartaric acid; L -(+)-Tartaric acid [CAS 526-83-0] and [CAS 87-69-4].
Tartaric Acid, dried over phosphorus pentoxide for 3 hours, contains not less than 99.7 percent and not more than 100.5 percent of C4H6O6.
Identification:
A: It responds to the tests for Tartrate.
B: When ignited, it gradually decomposes, emitting an odor resembling that of burning sugar
(distinction from citric acid).
Specific rotation: between +12.0 and +13.0 .
Test solution: 200 mg per mL, in water.
Loss on drying: Dry it over phosphorus pentoxide for 3 hours: it loses not more than 0.5% of its weight.
Residue on ignition: not more than 0.1%.
Limit of oxalate: Nearly neutralize 10 mL of a solution of it (1 in 10) with 6 N ammonium hydroxide, and add 10 mL of calcium sulfate TS: no turbidity is produced.
Sulfate: To 10 mL of a solution (1 in 100) add 3 drops of hydrochloric acid and 1 mL of barium chloride: no turbidity is produced.
Heavy metals: 0.001%.
Assay: Place about 2 g of Tartaric Acid, previously dried and accurately weighed, in a conical flask. Dissolve it in 40 mL of water, add phenolphthalein TS, and titrate with 1 N sodium hydroxide VS. Each mL of 1 N sodium hydroxide is equivalent to 75.04 mg of C4H6O6.
Tartaric Acid FCC Food Grade Specifications
L(+)-Tartaric Acid
C4H6O6 Formula wt 150.09
CAS: [87-69-4]
FEMA: 3044
DESCRIPTION
Tartaric Acid occurs as colorless or translucent crystals or as a white, fine to granular, crystalline powder. It is stable in air. One gram dissolves in 0.8 mL of water at 25C, in about 0.5 mL of boiling water, and in about 3 mL of alcohol. Its solutions are dextrorotatory.
Function: Acidifier; sequestrant; flavoring agent.
REQUIREMENTS
Identification: A sample solution gives positive tests for Tartrate.
Assay: Not less than 99.7% and not more than 100.5% of C4H6O6 after drying.
Lead: Not more than 2 mg/kg.
Loss on Drying: Not more than 0.5%.
Optical (Specific) Rotation: Between +12.0° and +13.0°.
Oxalate: Passes test.
Residue on Ignition: Not more than 0.05%.
Sulfate: Passes test.
Tartaric Acid Analytical Reagent Grade Specifications
2,3-Dihydroxybutanedioic Acid
HOOC(CHOH)2COOH
Formula Weight 150.0
CAS Number 87-69-4
REQUIREMENTS
Assay: Not less than 99.0%
MAXIMUM ALLOWABLE
Insoluble matter: 0.005%
Residue after ignition: 0.02%
Chloride (Cl): 0.001%
Oxalate (C2O4): Passes test
Phosphate (PO4): 0.001%
Sulfur compounds (as SO4): 0.002%
Heavy metals (as Pb): 5 ppm
Iron (Fe): 5 ppm.
We also manufacture Tartaric Acid EP JP IP Grade.
For Original Monographs of IP Indian Pharmacopoeia BP British Pharmacopoeia USP US Pharmacopoeia JP Japan Pharmacopoeia FCC Food Grade product, please check with the respective web-pages or books.
We manufacture and supply as under:
Ammonium Tartrate Dibasic or Diammonium Tartrate
Potassium Bitartrate, Cream of Tartar
Suberic Acid or Octanedioic Acid
N-Valeric Acid or N-Pentanoic Acid.
Manufacturers:
MUBY CHEMICALS
Ambernath Mumbai, Ankleshwar Gujarat, India
TEL: (OFFICE) +912223770100, +912223726950
Current Date Time in India GMT+5:30
e-mail: info@mubychem.com
USA, Canada, Mexico and other American
neighbouring buyers may
e-mail: us@mubychem.com
Call toll-free 1-877-682-9243 (1-877-MUBYCHEM)

Copyright and Usual Disclaimer is Applicable.
Last
21 January, 2025




Exporters to USA Canada UAE Europe South Africa Tanzania Kenya Uganda Egypt Nigeria Turkey Mexico Brazil Argentina Chile Dubai etc.
Global or International Suppliers, Exporters, Importers, Manufacturers
I shall pass through this world, but once. If therefore, there is any good that I can do, or if there is any favor that I can show to a fellow human being, let me do it now. Let me not defer or neglect it. For I shall not tread this way again
Tartaric Acid SDS, Safety Data Sheet
MSDS Sheet, Material Safety Data Sheet 21-Nov-20
1. Product Identification
Product Name & Other Names: Tartaric Acid or 2,3-Dihydroxybutanedioic acid
CAS No.: 87-69-4
EINECS EC Number: 201-766-0
Molecular Formula: C4H6O6
Molecular Weight: 150.1
Relevant uses and uses advised against (if any): Industrial Manufacturing.
Suppliers: As per letterhead.
2. Hazards Identification
GHS, Globally Harmonized System Classification in accordance with 29 CFR 1910
Classification according to Regulation (EC) No 1272/2008
Skin irritation Category 2, H315
Serious eye damage/eye irritation Category 2A, H319
Specific target organ toxicity - single exposure, Respiratory system. Category 3, H335
Labelling according Regulation (EC) No 1272/2008
GHS Label Elements  Irritant |
Signal Words: Warning
Hazard statements:
H315: Causes skin irritation.
H319: Causes serious eye irritation.
H335: May cause respiratory irritation.
Precautionary statements:
P264: Wash … thoroughly after handling.
P280: Wear protective gloves/protective clothing/eye protection/face protection.
P302+P352: IF ON SKIN: Wash with plenty of soap and water.
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing.
P332+P313: If skin irritation occurs: Get medical advice/attention.
P337+P313: If eye irritation persists: Get medical advice/ attention.
P362: Take off contaminated clothing and wash before reuse.
Classification according to EU Directives 67/548/EEC or 1999/45/EC:
Hazard Symbol:
Xi = Irritant.
Risk Phrases:
R36/37/38 Irritating to eyes, respiratory system, and skin.
3. Composition/Information on Ingredients
Product Name & Other Names: Tartaric Acid or 2,3-Dihydroxybutanedioic acid
CAS No.: 87-69-4
EINECS EC Number: 201-766-0
4. First Aid Measures
Always seek medical attention after first aid measures are provided.
Inhalation: If inhaled, remove to fresh air. If not breathing, give artificial respiration. If breathing is difficult, give oxygen and get medical attention immediately.
Ingestion: Never give anything by mouth to an unconscious person. Get medical attention.
Skin Contact: Immediately flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Get medical attention. Wash clothing and shoes before reuse.
Eye Contact: Immediately flush eyes with plenty of water for at least 15 minutes, lifting lower and upper eyelids occasionally. Get medical attention immediately.
5. Fire Fighting Measures
Fire: As with most organic solids, fire is possible at elevated temperatures or by contact with an ignition source.
Explosion: Fine dust dispersed in air in sufficient concentrations, and in the presence of an ignition source is a potential dust explosion hazard.
Fire Extinguishing Media: Water spray, dry chemical, alcohol foam, or carbon dioxide.
Flash point: 150C (302F) - closed cup.
Special Information: In the event of a fire, wear full protective clothing and NIOSH-approved self-contained breathing apparatus with full face piece operated in the pressure demand or other positive pressure mode. At high temperatures under fire conditions, it may produce toxic or irritating fumes. Fire-extinguishing work is done from the windward and the suitable fire-extinguishing method according to the surrounding situation is used. Uninvolved persons should evacuate to a safe place.
6. Accidental Release Measures
Personal precautions, protective equipment, and emergency procedures: Avoid breathing dust/fumes/gas/mist/vapors/spray. Use individual protective equipment (waterproof boots, suitable protective clothing, safety glasses, etc.). Restrict unprotected personnel from the area. Prevent any contact with hot surfaces. Do not approach facing the wind. Do not touch the spilled material.
Environmental precautions: Do not let the product enter drains, soil, or water sources.
Methods and materials used for containment Cleanup procedures and Storage:
Small Spill: Use appropriate tools to put the spilled solid in a convenient waste disposal container.
Large Spill: Do not inhale dust, vapors, mist, or gas. Avoid dust formation. Contain spilled material. Cover with an inert, non-combustible absorbent material, (e.g. sand, earth, diatomaceous earth, vermiculite). Use a shovel to put the material into a convenient waste disposal container. Finish cleaning by spreading water on the contaminated surface and allow to evacuate as per law.
7. Handling and Storage
Precautions for safe handling: Apply according to good manufacturing and industrial hygiene practices. Ensure proper ventilation. In case of insufficient ventilation, wear suitable respiratory equipment. Wash thoroughly after handling. Do not drink, eat, or smoke while handling. Avoid contact with skin, eyes, and clothing. Minimize dust generation. Avoid breathing dust/fumes/gas/mist/vapors/spray. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation. Use individual protective equipment (waterproof boots, suitable protective clothing, safety glasses, etc.). Prevent any contact with hot surfaces.
Conditions for safe storage, including any incompatibilities: Store in cool, dry, and ventilated area away from heat sources and protected from sunlight in tightly closed original container. Keep air contact to a minimum. Store protected from heat, sparks and ignition sources and incompatible materials. Avoid contact with skin and eyes. Avoid inhalation of dust/mist/vapor. Do not store with incompatible materials like strong oxidizing agents, bases, sodium nitrate, sodium nitrite, nitric acid. Avoid dust formation and control ignition sources. Employ grounding, venting and explosion relief provisions in accord with accepted engineering practices in any process capable of generating dust and/or static electricity.
8. Exposure Controls/Personal Protection
Airborne Exposure Limits: None established.
Ventilation System: A system of local and/or general exhaust is recommended to keep employee exposures as low as possible. Local exhaust ventilation is generally preferred because it can control the emissions of the contaminant at its source, preventing dispersion of it into the general work area. Please refer to the ACGIH document, Industrial Ventilation, A Manual of Recommended Practices, most recent edition, for details.
Personal Respirators (NIOSH Approved): For conditions of use where exposure to dust or mist is apparent and engineering controls are not feasible, a particulate respirator (NIOSH type N95 or better filters) may be worn. If oil particles (e.g. lubricants, cutting fluids, glycerin, etc.) are present, use a NIOSH type R or P filter. For emergencies or instances where the exposure levels are not known, use a full-face positive-pressure, air-supplied respirator. WARNING: Air-purifying respirators do not protect workers in oxygen-deficient atmospheres.
Skin Protection: Wear impervious protective clothing, including boots, gloves, lab coat, apron or coveralls, as appropriate, to prevent skin contact.
Eye Protection: Use chemical safety goggles and/or full face shield where dusting or splashing of solutions is possible. Maintain eye wash fountain and quick-drench facilities in work area.
Other Control Measures: Maintain good housekeeping in work area. Dust deposits on floors and other surfaces may pick up moisture and cause the surfaces to become slippery and present safety hazards. Handle in accordance with good industrial hygiene and safety practice. Wash hands after handling.
9. Physical and Chemical Properties
Appearance: White crystals or powder.
Odor: Not available.
Odor threshold: Not available.
pH: 1.0 - 2 at 150 g/l at 25C (77F).
Relative density: Not available.
Melting Point: 170C - 172C (338F - 342F) - lit.
Initial boiling point and boiling range: Not available.
Flash point: Not available.
Auto-ignition temperature: Not available.
Decomposition temperature: Not available.
Upper/lower flammability or explosive limits: Not available.
Vapor pressure: Not available.
Vapor Density (Air=1): 5.18
Evaporation rate: Not available.
Flammability (solid, gas): Not available.
Partition coefficient: n-octanol/water: Not available.
Solubility: Soluble in water. 150 g/l at 20C (68F)
Viscosity: Not available.
Molecular Formula: C6H4(CO2H)2
Molecular Weight: 150.1
10. Stability and Reactivity
Stability: Stable under ordinary conditions of use and storage.
Hazardous Decomposition Products: Carbon dioxide and carbon monoxide may form when heated to decomposition.
Hazardous Polymerization: Will not occur.
Incompatibilities: Strong oxidizers and bases, sodium nitrate, sodium nitrite, nitric acid.
Conditions to Avoid: Direct sunlight. Extremely high or low temperatures, Heat, dusting, and incompatibles.
11. Toxicological Information
LD50 oral rat: > 2000 mg/kg
LD50 dermal rat: > 2000 mg/kg
Lowest Published Lethal Dose LDLo, oral, rat: 7500 mg/kg
LD50 Intravenous - mouse - 485 mg/kg
Carcinogenicity: No component of this product present at levels greater than or equal to 0.1% is identified as possible or confirmed human carcinogen by IARC, ACGIH, OSHA and NTP.
Teratogenic Effects: Not available.
Mutagenic Effects: Not available.
Developmental Toxicity: Not available.
Reproductive Effects: No information available.
12. Ecological Information
EC50 Daphnia 1: 93.31 mg/l 48 hr.
EC50 other aquatic organisms 1: 51.4 mg/l 72 hr.
Environmental Fate: Sufficient information is not available. When released into the soil, Tartaric Acid is expected to leach into groundwater. When released into the soil, this material is expected to biodegrade. When released into water, this material is expected to biodegrade. This material is not expected to significantly bio accumulate.
Persistence and Degradability: Unlikely to persist due to water solubility.
Mobility: Likely to be mobile due to water solubility.
Bioaccumulation/ Accumulation: Not expected to significantly bioaccumulate.
Results of PBT and vPvB assessment: No data available for assessment.
13. Disposal Considerations
Whatever cannot be saved for recovery or recycling should be managed in an appropriate and approved waste disposal facility. Processing use or contamination of this product may change the waste management options. State and local disposal regulations may differ from federal disposal regulations. Dispose of container and unused Tartaric Acid in accordance with legal requirements.
14. Transport Information
DOT USA, TDG Canada & ADR/RID Europe: Not dangerous goods.
IMDG/IMO: Not dangerous goods.
IATA: Not dangerous goods.
15. Regulatory Information
USA:
SARA 302: Not subject to the reporting requirements of SARA Title III, Section 302.
SARA 313: Does not contain any chemical components with known CAS numbers that exceed the threshold (De Minimis) reporting levels established by SARA Title III, Section 313.
SARA 311/312 Hazards: Acute Health Hazard. See section 2.
California Prop. 65 Components: This product does not contain any chemicals known to State of California to cause cancer, birth defects, or any other reproductive harm.
Section 16 - Additional Information
European Labeling in Accordance with EC Directives:
H315 = Causes skin irritation.
H319 = Causes serious eye irritation.
H335 = May cause respiratory irritation.
Classification according to EU Directives 67/548/EEC or 1999/45/EC:
Hazard Symbol:
Xi = Irritant.
Risk Phrases:
R36/37/38 Irritating to eyes, respiratory system, and skin.
Disclaimer:
*****************************
Our company provides this MSDS sheet in good faith but makes no representation as to its comprehensiveness or accuracy. This SDS sheet is intended only as a guide to the appropriate precautionary handling of the material by a properly trained person using this product. The above information has been compiled from various sources and has the possibility of discrepancy and being out-dated information. Individuals receiving the information must exercise their independent judgment and do further search in determining its appropriateness for a particular purpose. In no case shall our company be liable to loss or damages by the product user.
*****************************















